Aging and stem cells
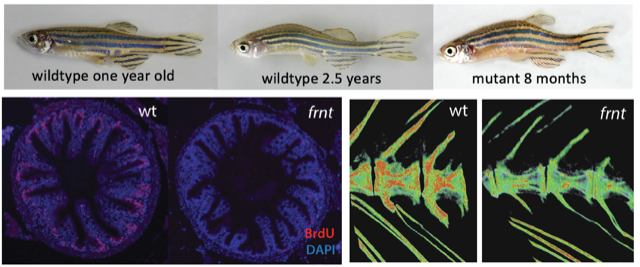
Zebrafish provide a unique model to study aging in vertebrates. As proof of concept, we identified mutants that showed progeric phenotypes, or advanced aging (above). These mutants showed all the hallmarks of normal vertebrate aging but at young ages. We identified the genetic cause of the mutant aging phenotype having novel function in regulating stem cell maintenance. Additionally, we have identified mutants affecting metabolic regulation of senescence as well as telomere function which can aid in our analysis of factors that may alleviate these age-related pathologies associated with these mutants. Current work is centered on identifying populations of postnatal stem cells and understanding signals to maintain their function in later life.
Publications
- Castro J., Fagerstrom, K., Treaster S., Duryea J., Charles J., Harris MP. The function and regulation of celsr1a in skeletal age-associated homeostasis and repair. Journal of Gerontology: Biological Sciences (In Press-2025)
- Li C, Barton C, Henke K, Daane J, Treaster S, Caetano-Lopes J, Tanguay R, and Harris MP. (2020). Celsr1a is essential for tissue homeostasis and onset of aging phenotypes in the zebrafish. eLife Jan 27;9. pii: e50523. PMID:31985398